In geology and astronomy, the term silicate is used to mean silicate minerals, ionic solids with silicate anions as well as rock types that consist predominantly of such minerals. Silicate anions are often large polymeric molecules with an extense variety of structures, including chains and rings (as in polymeric metasilicate [ SiO 2− The name is also used for any salt of such anions, such as sodium metasilicate or any ester containing the corresponding chemical group, such as tetramethyl orthosilicate. The family includes orthosilicate SiO 4−ħ ( x = 0.5, n = 2). In chemistry, a silicate is any member of a family of anions consisting of silicon and oxygen, usually with the general formula [ SiO (4−2 x)− It is not flammable.Structure of the orthosilicate anion SiO 4− This chemical compound may produce cancer by prolonged exposure. It can also cause respiratory irritation. Health effects/safety hazards: Silicon dioxide can cause serious eye irritation or damage. Silicon dioxide is extensively used as precursor to obtain glass and silicon, through the reaction: SiO 2 is the main component in the optical fibers for telecommunication. In pharmaceutical industries as additive of food and medicines to absorb water. It is also used to produce agricultural chemicals. In chemical industry, in the production of adhesives and sealants, adsorbents and absorbents, ceramic, porcelain, anti-adhesives, corrosion inhibitors, dyes and paint additives. Uses: Silicon dioxide has many uses in chemical, electronic and pharmaceutical industries. Moreover, it has a high dielectric strength, thus it is very used as insulator and semiconductor. The Si forms two double bonds with the oxygen, thus it is a very stable molecule. It is soluble in hydrofluoric acid.Ĭhemical properties: Silicon dioxide is not very reactive compound due the polarity of molecule is zero. Its melting and boiling point are 1600 ✬ and 2230 ✬, respectively. Physical properties: Silicon dioxide is a transparent to gray, odorless, crystalline or amorphous solid. The products are fine particles aggregates of 100 - 400 nm in diameter. Other methods let the production of pyrogenic silica and it consists on the combustion of silanes such as silicon tetrachloride, in an oxygen-hydrogen burner. Na 2Si 3O 7 + H 2SO 4 → 3 SiO 2 + Na 2SO 4 + H 2O This method is known as wet process and produces amorphous SiO 2 particles. Preparation: Although most of silicon dioxide is extracted through quartz mining, it can also be prepared through the acid neutralization of an aqueous alkali metal silicate solution. It is also present in the composition of many plants and in consequence, it can also be ingested by herbivores. It is the main component of mineral such as quartz, tridymite, amethyst, cristobalite, agateor sand. Occurrence: Silicon dioxide is a compound largely found in nature.
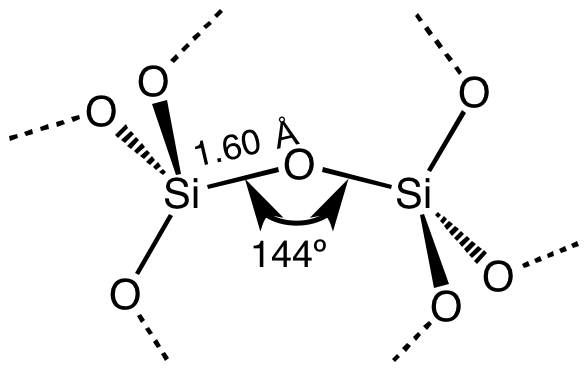
Its chemical structure can be written as below, in the common representations used for organic molecules. SiO 2 can also be found as an amorphous solid or other crystalline forms as rhombohedral, hexagonal, cubic, nonclinical or ortorhombic geometry. The compound is mostly found in the crystalline form, for example as quartz, which is formed by a cation Si central, coordinated to 4 anion O, so that the structure of silica has a tetrahedral geometry. Silicon dioxide is an oxide formed by Si +4 and O -2. Formula and structure: The silicon dioxide chemical formula is SiO 2.
